|
To receive all DailyMed Updates for the last seven days Labeler - Zydus Pharmaceuticals (USA) Inc. SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2)ģ0 in 1 BOTTLE Type 0: Not a Combination Productĩ0 in 1 BOTTLE Type 0: Not a Combination Productġ00 in 1 BOTTLE Type 0: Not a Combination Productġ000 in 1 BOTTLE Type 0: Not a Combination Productġ0 in 1 BLISTER PACK Type 0: Not a Combination Product MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) MECLIZINE HYDROCHLORIDE (UNII: HDP7W44CIO) (MECLIZINE - UNII:3L5TQ84570) Inform patients that alcohol may increase adverse reactions.Īdvise patients to notify their healthcare provider about all of their medical conditions, including if they are pregnant or plan to become pregnant or if they are breastfeeding. Inform patients that meclizine hydrochloride may impair their ability to engage in potentially dangerous activities, such as operating machinery or vehicles.Īdvise patients regarding medications that should not be taken in combination with meclizine hydrochloride or that may necessitate increased monitoring.  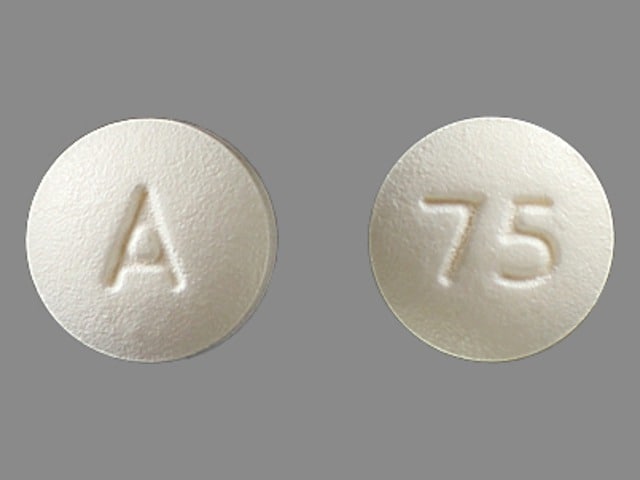
NDC 70710-1162-4 in unit-dose blister cartons of 100 tablets (10 X 10 Unit-dose)Īdvise patients that the tablets must be swallowed whole.Īdvise patients that meclizine hydrochloride may cause anaphylactic reaction, drowsiness, dry mouth, headache, fatigue, vomiting and, on rare occasions, blurred vision. NDC 70710-1162-0 in bottles of 1000 tablets with non-child-resistance closure NDC 70710-1162-1 in bottles of 100 tablets with child-resistance closure NDC 70710-1162-9 in bottles of 90 tablets with child-resistance closure NDC 70710-1162-3 in bottles of 30 tablets with child-resistance closure Meclizine Hydrochloride Tablets USP, 25 mg are light yellow to yellow colored, spotted, oval shaped, biconvex uncoated tablets debossed with '1162' on one side and plain on the other side and are supplied as follows: NDC 70710-1161-4 in unit-dose blister cartons of 100 tablets (10 X 10 Unit-dose) NDC 70710-1161-0 in bottles of 1000 tablets with non-child-resistance closure NDC 70710-1161-1 in bottles of 100 tablets with child-resistance closure NDC 70710-1161-9 in bottles of 90 tablets with child-resistance closure NDC 70710-1161-3 in bottles of 30 tablets with child-resistance closure Meclizine Hydrochloride Tablets USP, 12.5 mg are light blue to blue colored, spotted, oval shaped, biconvex uncoated tablet, debossed with '1161' on one side and plain on the other side and are supplied as follows: These effects occurred at doses as low as 25 mg/kg, which is approximately 2 times the maximum recommended human dose (100 mg) on a body surface area (mg/m 2) basis. In a published study, oral administration of meclizine (25 mg/kg to 250 mg/kg) to pregnant rats during the period of organogenesis resulted in a high incidence of fetal malformations. 
The background risk of major birth defects and miscarriage for the indicated population is unknown.Įpidemiological studies reporting on pregnancies exposed to meclizine have not identified an association between the use of meclizine during pregnancy and an increased risk of major birth defects. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively. However, in a published study, an increased incidence of fetal malformations was observed following oral administration of meclizine to pregnant rats during the period of organogenesis, at doses similar to those used clinically. Data from epidemiological studies have not generally indicated a drug-associated risk of major birth defects with meclizine during pregnancy.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
